A centralized design pairs high‑density sludge neutralization with a staged precipitation train — and finishes with membrane or ion‑exchange polishing — to meet very low discharge limits.
Industry: Nickel_Mining | Process: Wastewater_Treatment
Nickel miners are under pressure to move billions of liters of acidic, metal‑rich water into compliance with tight local standards. One route that has quietly become the industry’s workhorse: an HDS (high‑density sludge) lime process up front for pH neutralization and heavy‑metal precipitation, followed by multi‑stage chemical precipitation to selectively remove metals, and a final polishing step for sub‑ppm discharge.
It is not just chemistry. It is density, recycle, and separation design — and it is already proven at scale. Commercial HDS plants routinely show >90–99% dissolved‑metal removal when pH is controlled, with sludges that dewater and store better than conventional lime slurries (mdpi.com; sgs.com).
HDS neutralization and sludge densification
In HDS, ground limestone (CaCO₃)/dolomite and slaked lime (Ca(OH)₂) are added along with recycled sludge in a high‑shear, aerated reactor; the recycled sludge provides nucleation sites for precipitation, aggressive aeration oxidizes Fe²⁺ to Fe³⁺, and the rising pH (often toward 8–10) hydrolyzes dissolved metals. The result is copious precipitation of gypsum (CaSO₄·2H₂O), iron/manganese hydroxides/oxides, and heavy metal hydroxides (mdpi.com).
Sukati et al. report HDS sludges routinely contain 15–70% solids (dry bulk 1.05–1.37 g/cm³), versus only ~5% for conventional lime sludges (mdpi.com). SGS notes that by raising sludge solids from ~2% to ~30%, sludge volume is cut by >95%, and within days the sludge “drains” to >50% solids and is mechanically stable (supporting foot traffic) (sgs.com; sgs.com). For scale, “20 ton HDS per ML AMD” is cited in coalfields — thousands of tons daily — so volume reduction is critical (mdpi.com).
Besides physical benefits, recycled‑seeding imparts chemical stability: HDS effluents historically show minimal leaching, limiting groundwater contamination (sgs.com; mdpi.com). Commercial HDS plants (e.g., South African goldfields) routinely achieve >90–99% removal of dissolved metals (Fe, Al, Mn, Ni, Co, etc.) with pH control and reactors run to pH ≈9–10.5 so most transition metals precipitate as hydroxides (mdpi.com; sgs.com). SGS reports that after HDS, effluent quality normally meets or exceeds regulatory limits even for stringent local standards (sgs.com).
Design‑wise, plants allocate reactors (lime‑mix and aeration), a flocculation/clarification system, and return thickener underflow to the mix tank; typical recycle ratios are ~0.5–1.0× flow, and lime addition can run ~8–15 kg Ca(OH)₂/m³ effluent depending on acidity (mdpi.com; nepis.epa.gov). Stable gypsum/oxide sludges minimize risk and meet classification standards (often non‑hazardous) (mdpi.com). To execute the chemistry consistently, plants specify metering like a dosing pump for lime, oxidants, and sulfide feeds, and use a gravity separator such as a clarifier; for tight pads, a lamella settler reduces footprint by up to 80% compared to conventional clarifiers.
Multi‑stage precipitation train
Different metals have distinct pH‑solubility profiles, so dividing treatment into controlled stages maximizes total removal. One study achieved ~99.8% Fe removal and >94% co‑precipitation of Cu/Zn using staged oxidation and pH steps — suggesting Ni/Co would behave similarly at high pH (researchgate.net).
Stage 1 (iron/aluminum hydroxides): after initial neutralization, adjust pH to ≈3.5–5 to precipitate Fe and Al (as Fe(OH)₃/Al(OH)₃). A small H₂O₂ dose oxidizes Fe²⁺→Fe³⁺; the goal is to remove >90% of Fe/Al, which co‑precipitate many co‑contaminants. Wang & Chen (2019) reported >99% Fe removal at pH ~3.5–4 with a small H₂O₂ dose, and many other metals co‑precipitate at this step (for example, 95% of Zn can co‑precipitate by pH 6–8) (researchgate.net).
Stage 2 (sulfide precipitation): add a soluble sulfide (e.g., NaHS/Na₂S) or generate H₂S gas with strict pH control to precipitate remaining soft metals as sulfides. Under neutral to slightly alkaline conditions (pH ~7–9), metals such as Cu, Ni, Co, Pb, Zn form low‑solubility sulfides (e.g., NiS, Cu₂S). Because metal sulfides have much lower Ksp than hydroxides, this polishes residual parts per million; adding NaHS after initial precipitation has removed >90% of Cu and Zn in testing, with similar efficiency expected for Ni/Co. Practically, one study fed 1 g/L NaHS at pH ~3.5–4 then raised pH to 9, recovering ~94% Cu and 96% Zn (researchgate.net). NiS solubility is ~10⁻²¹, far below Ni(OH)₂’s ~10⁻¹⁴, so sulfide every drop if needed.
Stage 3 (final hydroxide precipitation): raise pH to ~9–10 with Ca(OH)₂ to precipitate any remaining metal hydroxides (Ni(OH)₂, Co(OH)₂, Zn(OH)₂, etc.). A final MgO or Ca(OH)₂ dose may be used; any residual iron/chrome co‑precipitates. In practice, these sequential steps achieve >90–99% cumulative recovery of each metal; one integrated process achieved 99.8% Fe, 94.0% Cu, and 96.1% Zn removal (researchgate.net). Each step uses simple gravity separation (clarifiers or thickeners) to draw off sludge, which can be recycled HDS‑style to seed crystallization and increase density.
Final polishing to very low limits
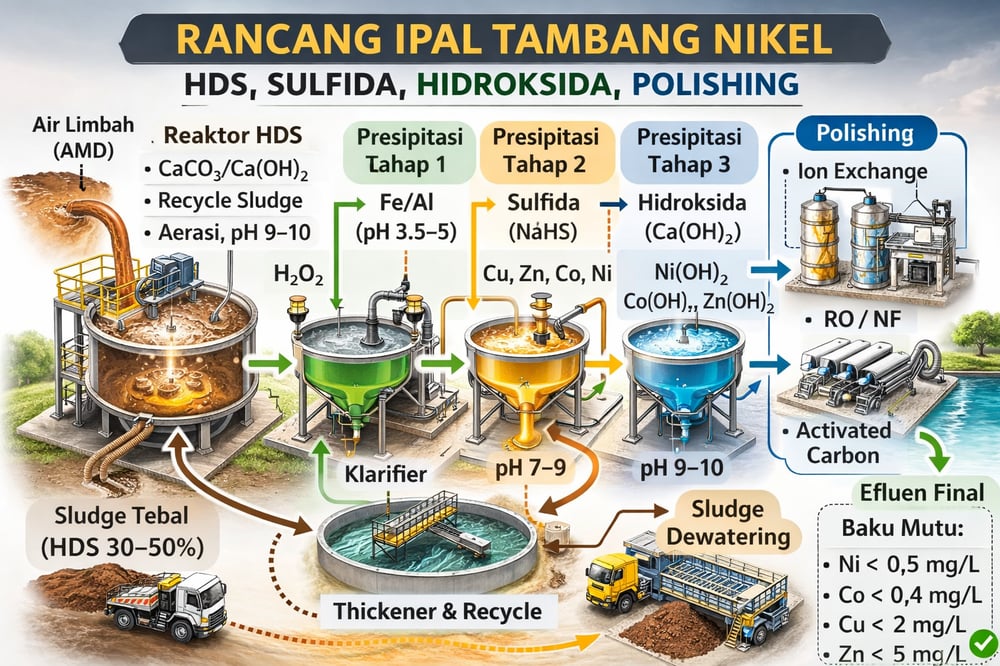
After Stage 3, effluent is largely cleared of target metals; a residue containing <0.1–0.5 mg/L Ni is typical, depending on starting load. Indonesian standards (Permen LH 09/2006) set Ni ≤0.5 mg/L, Co ≤0.4 mg/L, Cu ≤2 mg/L, Zn ≤5 mg/L for Ni‑ore mining effluent (ro.scribd.com), and many sites discharge to the sea or strict‑regulated rivers. A final polishing step is therefore routine.
Ion exchange: strong‑base cation exchange resins can adsorb residual Ni, Co and other divalent cations; historical data show resins can remove Ni to <0.4 mg/L in treated effluent (nepis.epa.gov). Resins selectively capture the last fractions, then are regenerated, producing a small, concentrated brine for disposal or recovery. For packaged or permanent trains, operators deploy ion‑exchange systems and match media using ion‑exchange resin suited to Ni/Co polishing.
Membranes: pressure‑driven membranes (nanofiltration, NF; reverse osmosis, RO) reject dissolved salts and metals. NF/RO is common in metal finishing to produce near‑zero‑metal effluent (mdpi.com); ultrafiltration (UF) or nanofiltration has reduced Ni to essentially zero in electroplating wastewater (mdpi.com). In practice, an RO stage after precipitation yields a high‑quality permeate (<μg/L metals) and a small concentrate; the concentrate can be recycled to precipitation or handled separately. To configure RO, NF, and UF as an integrated suite — “RO, NF, and UF systems for industrial and municipal water treatment” — operators combine nano‑filtration and brackish‑water RO, with ultrafiltration as pretreatment. Membrane polishing is costly and energy‑intensive but guarantees compliance with strict limits (often <0.01 mg/L for potable reuse) (mdpi.com).
Adsorption/advanced oxidation: in some designs, activated carbon or proprietary metal‑scavenging media (iron oxyhydroxides, bone‑char, polymer‑chelates) are used in final clarification to “mop up” trace ions; these have high affinity for Ni²⁺ at neutral pH. Natural clays or iron‑based adsorbents have been tested to bring Ni down to sub‑0.1 mg/L, though capacities vary (mdpi.com; nepis.epa.gov). Where adsorption makes sense, plants commonly specify an activated carbon filter as the polishing bed.
Compliance benchmarks and equipment choices
Because Indonesian nickel operations often discharge to the sea or strict‑regulated rivers, final effluent is expected to be essentially metal‑free (or at least below 0.5 mg/L Ni). Many international plants combine polishing — for example, an ion‑exchange train followed by RO — and the U.S. EPA (1980) notes that combined precipitation plus polishing (ion‑exchange or RO) is now standard for smelter/semi‑metals effluents (nepis.epa.gov; nepis.epa.gov). In practice, the final step is chosen by testwork: if residual Ni is ~0.2 mg/L after Stage 3, a resin column can lower it to <0.05 mg/L. Process performance is measured (and often automated) to ensure discharge targets are met at <100% of limit.
Summing the mass balance: if feed Ni is 100 mg/L, staged precipitation can reduce it to ~1–2 mg/L, and polishing to <0.5 mg/L or lower. Sludge solids can reach ~30–50% by dry weight, shrinking disposal volume by ~90+% (sgs.com). Compliance is demonstrated via regular effluent monitoring; Indonesian MOEF standards and local government may enforce Ni ≤0.5 mg/L, Co ≤0.4 mg/L (ro.scribd.com). For packaged integration of RO, NF, and UF, many operators standardize on membrane systems and support gear such as water‑treatment ancillaries to ensure stable operation.
Citations and performance references
Evidence comes from recent reviews and case studies: SGS reports on large‑scale HDS projects (sgs.com), and Sukati et al. (2021) analyze HDS sludge chemistry (mdpi.com). A 2019 ASCE paper details sequential precipitation of Fe, Cu, Zn with >94% recovery (researchgate.net). U.S. EPA guidelines (1980) describe typical designs (lime precipitation, ion exchange, RO) (nepis.epa.gov; nepis.epa.gov). Indonesian Reg. No. 09/2006 gives pollutant limits (ro.scribd.com). Membrane and polishing methods are documented in recent literature (mdpi.com; nepis.epa.gov). Amped with these data‑backed designs and separations, a new Ni‑mine wastewater plant can assure regulators — and shareholders — of sustainable, cost‑effective heavy‑metal treatment.
