Automotive pretreatment and electrophoretic coating wastewater is a complex cocktail of metals, phosphates, oils, and surfactants. A centralized treatment design—equalization, chrome reduction, multistage precipitation, and clarification—has emerged as the pragmatic, regulator‑proof playbook.
Industry: Automotive | Process: Pre
Automotive pretreatment/e‑coat lines shed more than rinse water. Industry studies flag high suspended solids, oils, surfactants, organics (chemical oxygen demand/biological oxygen demand, indicators of organic load), phosphates from Zn/Mn phosphating, and heavy metals including chromium, nickel, zinc, and copper (researchgate.net). Surveys and pilots report metals in the tens of mg/L (Ni ~5–30 mg/L, Zn up to ~100–150 mg/L) and phosphate (~20–50 mg/L) (researchgate.net) (researchgate.net).
Regulatory lines are tight. Indonesia’s PermenLH No.69/2013 caps hexavalent chromium at 0.1 mg/L (total Cr ~0.5 mg/L in strict zones) (scribd.com), while similar global regimes (EPA, CPCB) constrain Cd, Ni, Zn often to much less than 1 mg/L. The chemistry can deliver: Liu et al. reported ~96.6% phosphate removal and ~98% nickel removal at pH ~10, implying residual PO₄ at ≲1–2 mg/L (researchgate.net). A 110 kL/d effluent plant in India has demonstrated compliant operation under CPCB oversight (researchgate.net).
Centralized treatment train design
The recommended sequence is: (1) coarse pre‑treatment (screening/degreaser) and oil‑water separation if needed, (2) equalization basin, (3) chromium(VI) reduction reactor, (4) high‑pH, multistage precipitation/coagulation, and (5) final clarification. Each stage is sized for peak flow and pollutant load, with controls to stabilize reaction conditions (researchgate.net). Physical front‑end equipment, such as waste‑water physical separation systems and targeted oil removal, reduces load on downstream chemistry.
Automated chemical addition underpins the flow sheet. Controlled pH shifts, reduction, coagulation, and flocculation are maintained via feedback to metering equipment; industrial plants commonly deploy a dosing pump for proportional chemical feed tied to sensor readings.
Equalization tank sizing and control
A well‑mixed equalization (EQ) tank with 8–24 hours of retention evens out intermittent discharges and buffers pH swings. Typical design targets include handling ±30–100% load swings and sizing volume to approximately 1–2 days of average flow, with mechanical mixing to keep wastewater homogeneous. Online pH measurement and level control are critical; the EQ tank protects downstream basins from shock loads such as high COD or low‑pH spikes (researchgate.net).
Hexavalent chromium reduction reactor
Where chromate rinses contribute Cr(VI), part or all EQ effluent is routed through a dedicated reduction step before neutralization. A reducing agent—sodium bisulfite (NaHSO₃) or a ferrous salt—is dosed at low pH (~2–4) to convert Cr⁶⁺ to Cr³⁺, with typical stoichiometry calling for a few grams of NaHSO₃ per mg of Cr(VI). Online ORP (oxidation‑reduction potential) and pH monitoring, or Cr⁶⁺ dip tests, verify >95% reduction in a 10–30 minute, well‑mixed reactor. Iron‑based reduction has taken Cr/Ni/Cu down to ~0.1 ppm in plating wastewater (researchgate.net).
Multistage chemical precipitation chemistry
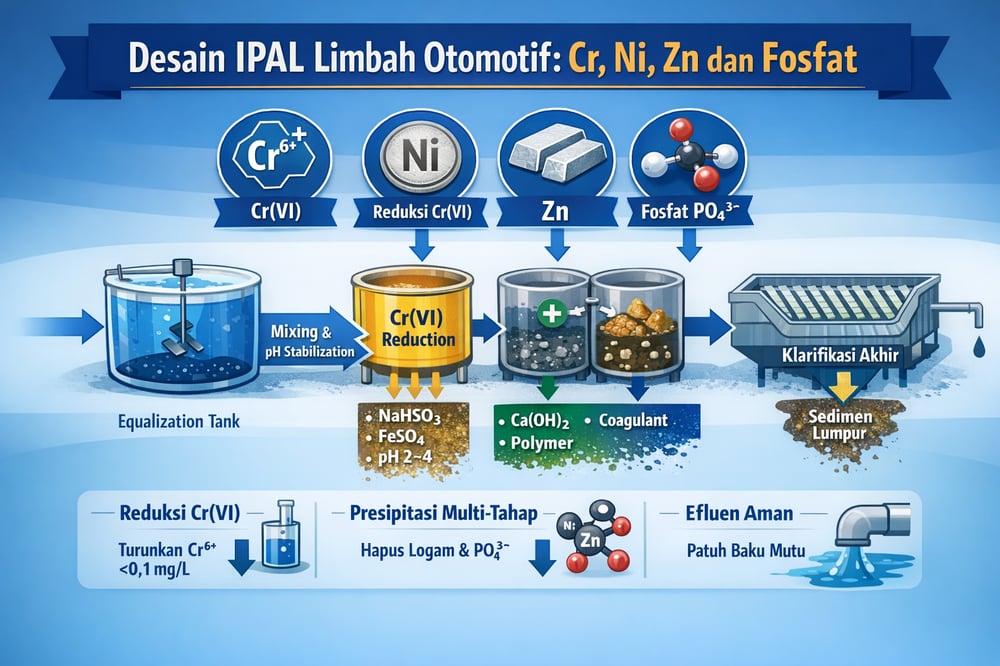
After reduction, the combined flow is raised to high pH (typically 9–10) with lime (Ca(OH)₂) or NaOH in a reaction/coagulation basin. At pH ≈9–10, heavy metals such as Ni²⁺, Zn²⁺, Cu²⁺, Cr³⁺, and Mn²⁺ precipitate as hydroxides (e.g., Zn(OH)₂, Ni(OH)₂) (wxyosun.com) (researchgate.net). Removal of Cu and Zn above 90% at pH 8–10 is documented (researchgate.net).
In Stage 1 (alkaline precipitation), the setpoint is raised to pH 9.0–9.5 and a coagulant salt, such as ferric chloride or polyaluminum chloride (PAC), is introduced. Ferric (Fe³⁺) or aluminum (Al³⁺) aids floc formation and co‑precipitates phosphate as FePO₄; calcium can yield hydroxyapatite (Ca₅(PO₄)₃OH) and aluminum phosphate (AlPO₄) (wxyosun.com). Empirically, Fe/P or Al/P molar ratios near 2:1 are applied to remove ≥90% of phosphate across ~20–50 mg/L feeds, and Liu et al. reported that 0.5 g/L PAC at pH ~10 removed ~96.6% of total phosphorus (researchgate.net). Plants often specify PAC as a standard coagulant; a commercial option is listed under PAC coagulants.
Polymer flocculation and mixing
Following precipitation, a polymer flocculant—commonly a cationic polyacrylamide (PAM)—aggregates fine precipitates into settleable flocs. Gentle mixing in a flocculator for ~30–60 minutes promotes large, dense flocs that settle rapidly; jar tests determine optimal dose. Industrial formulary references include flocculants tailored for clarifier performance.
Final clarification and sludge handling
A clarifier—often a space‑efficient plate design—finishes solids separation. Lamella (plate) clarifiers are compact and achieve >90% suspended solids removal, and, with several hours of hydraulic retention, produce effluent total suspended solids below 50 mg/L, often below 10 mg/L. A purpose‑designed lamella settler reduces footprint relative to conventional basins while sustaining settling rates.
Settled sludge contains metal hydroxides (e.g., Zn/Cu hydroxides) mixed with iron/aluminum phosphate by‑products per the cited precipitation reactions (wxyosun.com). Classified as B3 (hazardous), this sludge is dewatered—filter presses typically produce cakes with 65–75% moisture (wxyosun.com)—and sent for licensed disposal or metal recovery in accordance with PermenLHK requirements.
Optional polishing steps
Where stringent COD or TSS targets apply, a downstream polish—sand‑carbon filtration or ultrafiltration—tightens effluent quality beyond clarification. Pretreatment‑grade membranes, such as ultrafiltration, provide a barrier for fine colloids and emulsified residuals ahead of discharge or reuse.
Operational monitoring and control
Continuous monitoring is central to compliance. Online pH and redox sensors at key points maintain pH ~2–4 in the Cr(VI) reduction reactor and pH 9.0–10.0 in the precipitation tank, with feedback control (proportional dosing) on chemical feeds. If outlet Cr⁶⁺ drifts above ~0.02–0.05 mg/L, control logic increases reductant flow; this tight control has delivered final Cr/Ni/Cu near 0.1 mg/L (researchgate.net). Regular calibration of sensors and chemical meters is part of the instrumentation plan; supporting panels and auxiliaries are available under wastewater ancillaries.
Sampling, optimization, and compliance
Composite effluent sampling (daily/weekly) for metals, phosphorus, pH, and COD enables trend analysis. Operational targets used by compliant facilities include heavy metals safely below limits (e.g., <0.05 mg/L Cr³⁺, <0.2 mg/L Ni, <0.5 mg/L Zn) and total phosphorus at <1–2 mg/L; deviations trigger process checks. A documented 110 kL/d plant maintained parameters within India’s CPCB requirements under these controls (researchgate.net).
Jar testing on equalized influent weekly or monthly refines chemical setpoints. For lime [Ca(OH)₂], removal gains level off beyond pH 10 (researchgate.net). Coagulant dose is verified with new chemical batches or if influent P/COD shifts; PAC or ferric chloride is adjusted to hold P‑removal above 95% (researchgate.net). Aluminum chlorohydrate is an alternative coagulant used in low‑pH or difficult waters and is available under ACH.
pH neutralization and discharge range
Acidic or alkaline slugs from upstream rinses are homogenized in the EQ tank, and final neutralization confirms the discharge pH band. Verification focuses not only on reactor setpoints but on decant pH meeting typical 6–9 limits.
Sludge management, safety, and maintenance
Clarifier sludge blanket depth is observed routinely; withdrawal is scheduled to avoid carry‑over. Sludges often consolidate rapidly—Liu et al. observed full settling in <20 minutes when magnetite was used in the coagulation train (researchgate.net). Daily records of sludge volumes flag overdosing or spills. Corrosive reagents (H₂SO₃, NaOH, FeCl₃) necessitate corrosion‑resistant piping and spill protocols, along with operator training and personal protective equipment; mixers, pumps, and chemical‑feed lines receive preventive maintenance.
Documentation and expected performance
Regulators (CPCB, MoEF) expect documented evidence of consistent performance: flow rates, chemical additions (kg/day), sample results, and maintenance logs. Facilities use these records to demonstrate efficiency; for example, “Ni was consistently reduced by >98% (to <0.1 mg/L) with 1 g/L PAC at pH 10” (researchgate.net). With the design and practices above, typical removal expectations are >95% for heavy metals—yielding final concentrations around ~0.1 mg/L or lower (researchgate.net) (researchgate.net)—and >90–96% for phosphates, to ~<1–2 mg/L (researchgate.net).
Sources: Authoritative studies and guidelines on metal finishing wastewater treatment—researchgate.net; researchgate.net; researchgate.net; researchgate.net; and regulatory references in scribd.com.
